REDUCTION OF EMISSIONS DURING START-UP
Many emissions are being generated during the initial hours of the start-up of the cell. A number of options are available to reduce these emissions:
- bake the cathode using specialised equipment
- replace the monolithic mix by prebaked blocks
- install a hood with ducts and a scrubber system
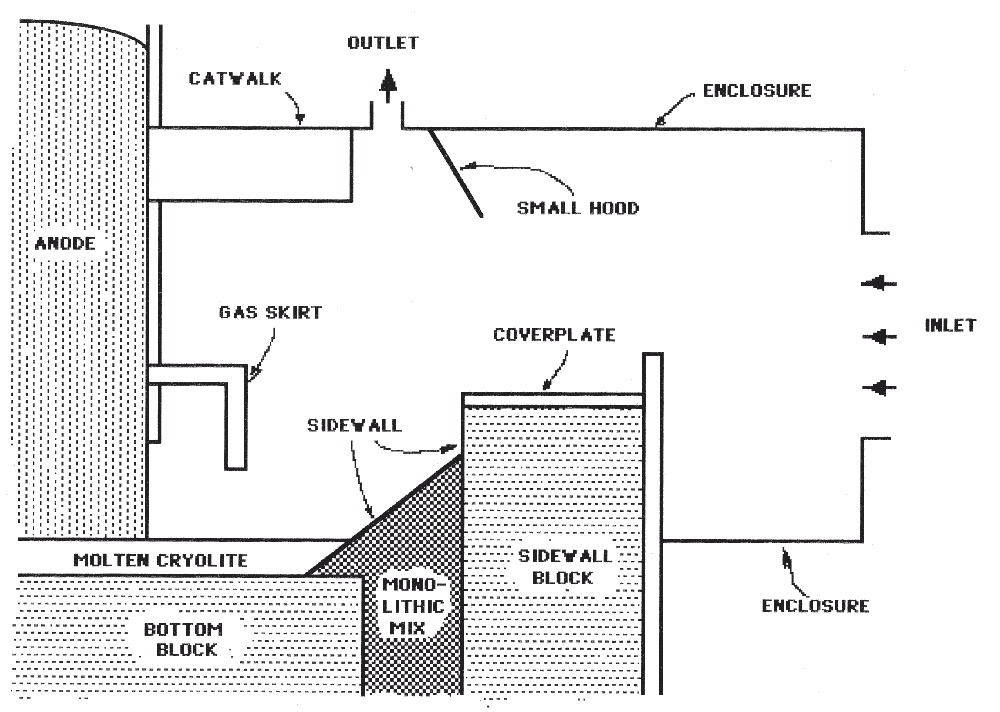
The first two options would address the hydrocarbon emission problem, but not the fluoride emission problem during start-up. Hence the present study focussed on the third option. A completely closed hood around the cell was not chosen because the high temperatures under the hood could damage some structural parts of the cell and because the operators need to have frequent access to the cell during the start-up. An open hood around the cell was therefore chosen. The VS Söderberg cell with an experimental hood is shown in Figure 1.
The required gas exhaust rate at the hood was estimated by flow calculations using the finite volume CFD code TASCflow3D of Advanced Scientific Computing (ASC). Two cases were studied: the capture of the hydrocarbon and fluoride fumes and the capture of the fluoride fumes.
MODEL GEOMETRY AND MODEL ASSUMPTIONS
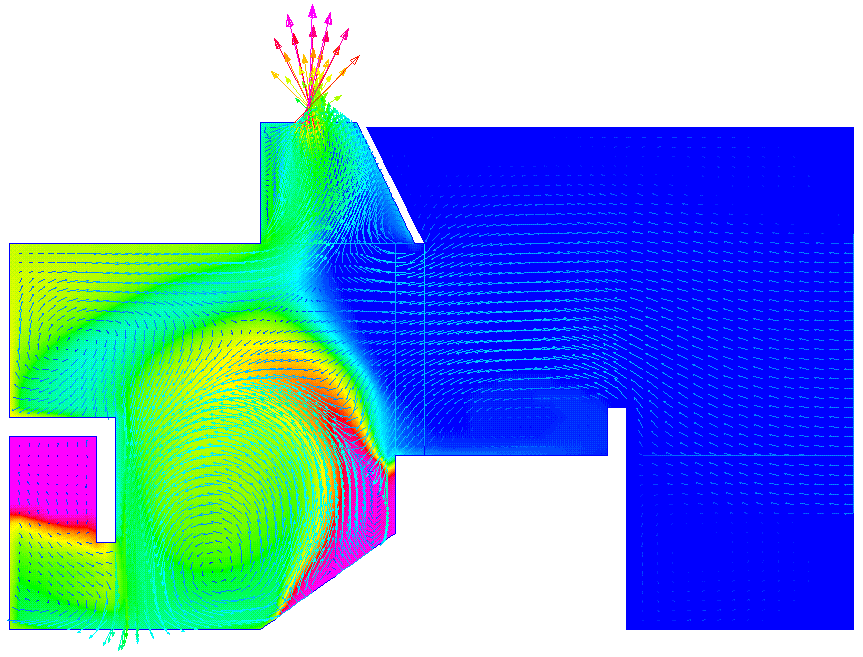
The VS Söderberg cell is about 30 feet long and 15 feet wide. In practice, the flow domain near the cell is open, but for the purpose of the simulation the domain was closed by an enclosure attached to the side of the Söderberg cell. The inlet opening to the enclosure was large in order to maintain a low flow velocity near the cell. The outlet opening was at the top of the small hood. Although there are some three dimensional flow effects, a two dimensional model was considered to be adequate for the present purpose. The model represented a vertical slice at full size near the side of the cell.
Following the recommendation of ASC, the depth of the model was represented by two layers and a symmetry boundary condition was applied to the
two end planes of the two layers. The problem was thus defined as a pseudo-2D problem [4]. The 2D flow domain was divided using a structured mesh and the multi-block feature. The model geometry is shown in Figure 2. About 3800 elements were used for the model.
Due to the hot surfaces the flow near the cell is induced by turbulent natural convection. Cool air at low velocity is sucked towards the cell. The effect of turbulence was modelled by increasing the laminar viscosity and the thermal conductivity by a factor of 20. Although the k-e model of turbulence was available in TASflow3D, previous work [5] has shown that the use of the k-e model gives no better results than the enhanced viscosity model. The Reynolds Flux turbulence model is best suited to simulate such a mixed flow system, but it was not available in TASCflow3D.
The solution was obtained by solving the Navier-Stokes and the energy equation simultaneously and activating the buoyant flow feature in TASCflow3D.
The time-averaged Navier-Stokes equation is usually written:
![]() (1)
(1)
The time-averaged energy equation is:
![]() (2)
(2)
For most of the surfaces of the flow domain, the velocities and heat fluxes were set to zero. A velocity and a temperature of 20°C were specified at the inlet opening. Zero pressure was specified at the outlet opening. The two flow cases were defined by particular boundary conditions on the hot surfaces of the flow domain.
The simulation runs were made by trial and error by specifying a velocity at the inlet opening. This velocity was decreased progressively until warm air started to spill from the hood and accumulated in the space at the top of the enclosure. This spill-over was unstable and varied with successive iterations. As the exhaust rate was decreased, the frequency of spill-overs from the hood was increased. The minimum required exhaust rate was thus the inlet flow which prevented any spill-over.